Factory best selling Fda Approved Cgm - UBREATH ® Spirometer System (PF280) – e-Linkcare
Factory best selling Fda Approved Cgm - UBREATH ® Spirometer System (PF280) – e-Linkcare Detail:

Reliable Result
Provides 6 parameters: PEF, FVC, FEV1, FEV1/FVC, EFE50, FEF75.
Accuracy and repeatability comply with ATS/ERS task force standardization (ISO26782:2009)
Complies with the ATS/ERS requirement for flow sensitivity down to 0.025L/s which is a vital characteristic for the diagnosis and monitoring of COPD patients.Portable Design
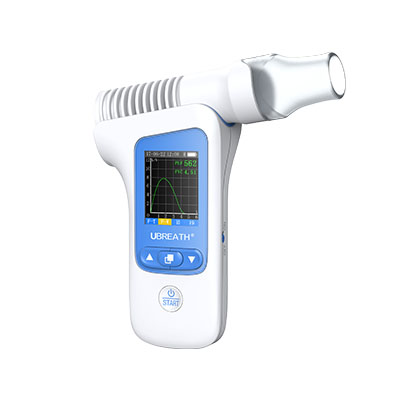
Hand-held device and easy to operation.
Automated BTPS calibration and free of environmental conditions influence.
Lightweight combines the benefits of portability.
Maintain easily and free daily Calibration.
Zero Cross-Contamination
Assured hygiene with disposable pneumotach gives NO authority to cross-contamination.
Patented design offers prevention and no daily sterilization is required.
Automated quality control and correction algorithm to minimize interference from operation.
User Friendly
Incentive graph and digital indicators displayed supports doctors’ disease quick assessment.
Colorful range indicator allows quick assessment for better visual clarity.
Easily connect to PC for data exchange.
NO daily Calibration and Sterilization required.
Data Transfer
Easily connect to PC via dedicated Bluetooth Communication Module for data exchange.
Access to UBREATH Software for more data analysis function.
UBREATH Spirometer System(Model No. PF280)is a high-quality, easy to use, portable spirometer that delivers an ideal combination of portability, accuracy and safety. And it also helps doctor analyze the pulmonary data through V-T/F-V curve and digital indicator which is an ideal solution for the primary care, point of care, patients self-monitoring environment.

Technical Specifications
|
Feature |
Specification |
| Model | PF280 |
| Parameter | PEF, FVC, FEV1, FEV1/FVC, FEF50, FEF75 |
| Flow Detection Principle | Pneumotachograph |
| Volume Range | Volume: 0.5-8 LFlow: 0-14 L/s |
| Performance Standard | ATS/ERS 2005 & ISO 26783:2009, ISO 23747:2015 |
| Volume Accuracy | ±3% or ±0.050L |
| Power Supply | 3.7 V lithium battery |
| Battery Life | Approximately 500 complete charge cycles |
| Printer | External Bluetooth printer |
| Memory | 495 records |
| Operating Temperature | 10℃ - 40℃ |
| Operating Relative Humidity | ≤ 80% |
| Size | Spirometer: 133x76x39 mm |
| Weight | 135g (including the Flow Transducer) |
Product detail pictures:
Related Product Guide:
The organization upholds the philosophy of "Be No.1 in good quality, be rooted on credit history and trustworthiness for growth", will keep on to provide previous and new customers from home and overseas whole-heatedly for Factory best selling Fda Approved Cgm - UBREATH ® Spirometer System (PF280) – e-Linkcare , The product will supply to all over the world, such as: Georgia, Indonesia, Guyana, Most problems between suppliers and clients are due to poor communication. Culturally, suppliers can be reluctant to question things they do not understand. We break down those barriers to ensure you get what you want to the level you expect, when you want it. Faster delivery time and the product you want is our Criterion .
After the signing of the contract, we received satisfactory goods in a short term, this is a commendable manufacturer.