OEM/ODM Manufacturer Impaired Glucose Tolerance Test - UBREATH ® Exhalation analyzer (BA200) – e-Linkcare
OEM/ODM Manufacturer Impaired Glucose Tolerance Test - UBREATH ® Exhalation analyzer (BA200) – e-Linkcare Detail:
Features:
Chronic airway inflammation is a general feature of some types of asthma, cystic fibrosis (CF), bronchopulmonary dysplasia (BPD), and chronic obstructive pulmonary disease (COPD).
In today’s world, a noninvasive,simple, repeatable, quick, convenient, and relatively low cost test called Fractional exhaled nitric oxide (FeNO), often plays a role to help identify airway inflammation, and thereby support a diagnosis of asthma when there is diagnostic uncertainty.
Fractional concentration of carbon monoxide in exhaled breath (FeCO), similar to FeNO, has been evaluated as a candidate breath biomarker of pathophysiological states, including smoking status, and inflammatory diseases of the lung and other organs.
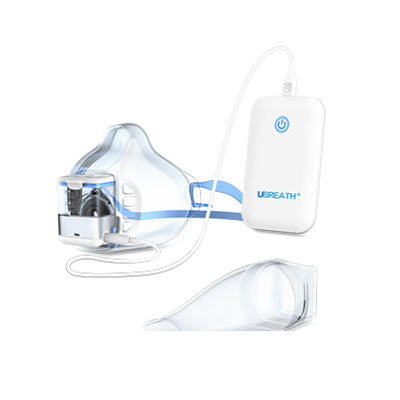
UBREATH Exhalation analyzer (BA810) is a medical device designed & manufactured by e-LinkCare Meditech to associate with both FeNO and FeCO testing to provide rapid, precise, quantitative measurement in order to assist with clinical diagnosis and management such as asthma and other chonic airway inflammations.
In today’s world, a noninvasive,simple, repeatable, quick, convenient, and relatively low cost test called Fractional exhaled nitric oxide (FeNO), often plays a role to help identify airway inflammation, and thereby support a diagnosis of asthma when there is diagnostic uncertainty.
| ITEM | Measurement | Reference |
| FeNO50 | Fixed Exhale flow level of 50ml/s | 5-15ppb |
| FeNO200 | Fixed Exhale flow level of 200ml/s | <10 ppb |
In the meantime, BA200 also provides data for the following parameters
| ITEM | Measurement | Reference |
| CaNO | Concentration of NO in the gas phase of the alveolar | <5 ppb |
| FnNO | Nasal nitric oxide | 250-500 ppb |
| FeCO | Fractional concentration of carbon monoxide in exhaled breath | 1-4ppm>6 ppm (if smoking) |
Product detail pictures:
Related Product Guide:
Our purpose would be to offer good quality products at competitive price ranges, and top-notch support to clients around the whole world. We're ISO9001, CE, and GS certified and strictly adhere to their good quality specifications for OEM/ODM Manufacturer Impaired Glucose Tolerance Test - UBREATH ® Exhalation analyzer (BA200) – e-Linkcare , The product will supply to all over the world, such as: Estonia, UK, Uruguay, Our company offers the full range from pre-sales to after-sales service, from product development to audit the use of maintenance, based on strong technical strength, superior product performance, reasonable prices and perfect service, we will continue to develop, to provide the high-quality products and services, and promote lasting cooperation with our customers, common development and create a better future.
This company can be well to meet our needs on product quantity and delivery time, so we always choose them when we have procurement requirements.